VetAlertTM African Swine Fever Virus (ASFV) DNA Test Kit
A Diagnostic PCR Test Kit for the Qualitative Detection of African Swine Fever Virus DNA extracted from spleen and whole blood collected in EDTA from swine. Availability subject to regulatory restrictions.
True Name: African Swine Fever Virus DNA Test Kit, Product Code 57A5.80
Catalog Number: TC-9096-096
USDA Licensed: 1 November 2021
Name and Intended Use
The ASFV DNA Test Kit is intended for the rapid in vitro qualitative detection of African Swine Fever (ASF) viral DNA by real-time polymerase chain reaction (rPCR). The reagents should be used to detect viral DNA extracted from spleen and whole blood collected in EDTA from swine. The test is intended for use by veterinary or other laboratory scientists for the presumptive identification of ASFV. The test should be performed on the ABI 7500 or an equivalent real-time PCR instrument.
Principles of the Procedure
The ASFV PCR test is a real-time PCR method that utilizes two independent, specific sets of forward and reverse primers, an inhibition control and fluorogenic probe hydrolysis chemistry for the detection of ASF viral DNA in samples. The test has specific sets of oligonucleotide primers and FAM probes that target the VP72 gene of the ASFV.
Sales and Use Restrictions
- Domestic distribution and use shall be under the supervision or control of USDA, APHIS, Veterinary Services, as part of an official USDA animal disease control program.
- Sale and use in the United States are restricted to laboratories approved by State and Federal (USDA) animal health officials.
- US laboratories authorized to perform ASFV testing are found on this list: https://www.aphis.usda.gov/animal_health/nahln/downloads/asf_lab_list.pdf
Limitations
This test is used for testing extracted DNA from spleen and whole blood samples collected in EDTA from swine. No claims are made for other sample types or for organisms derived from other animal species.
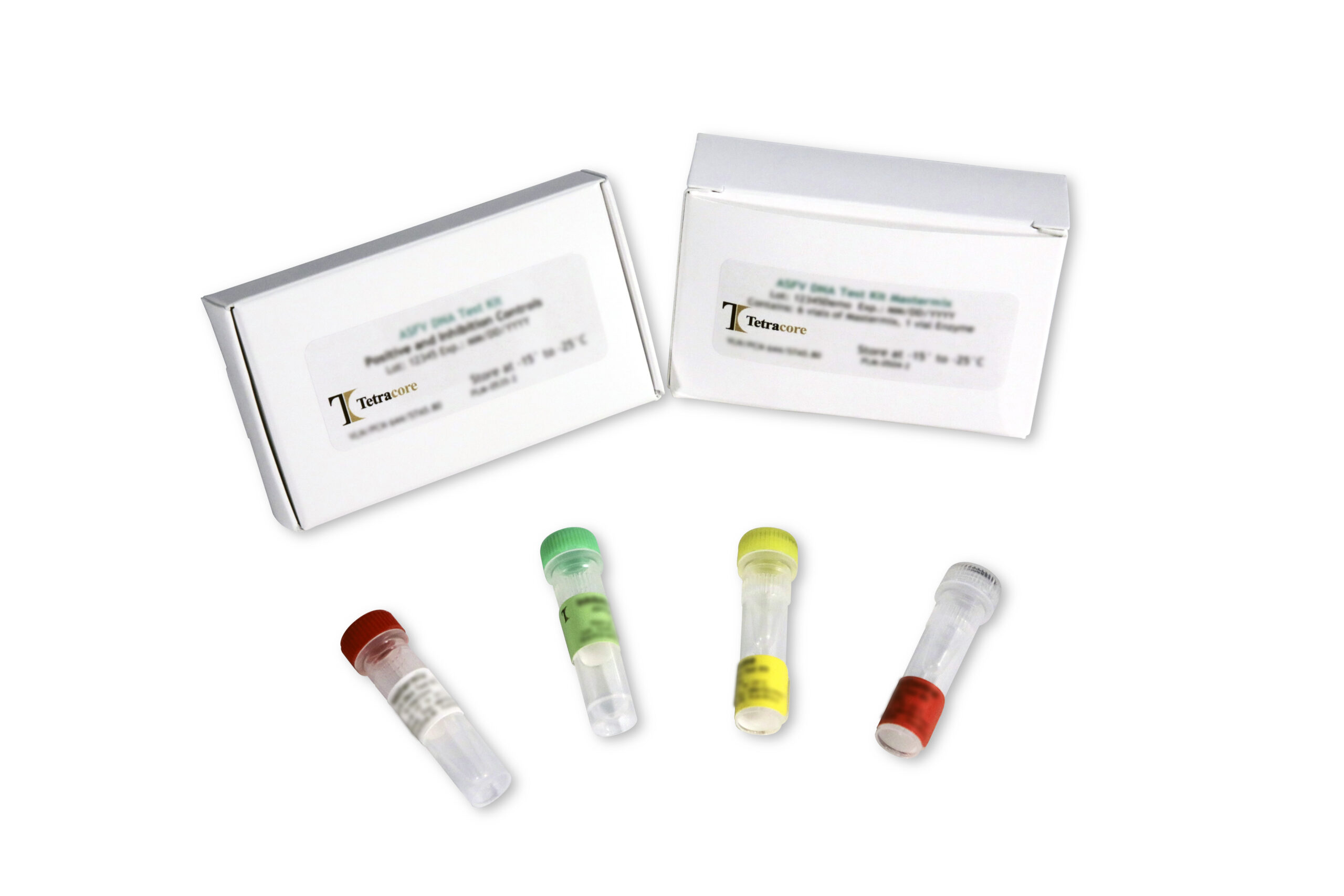
Materials Provided
| Component | Contents |
| Mastermix | 6 Tubes of ASF Mastermix
(16 reactions each) |
| Enzyme | 1 Tube of Enzyme |
| Positive Control | 1 Tube of ASF Positive Control |
| Inhibition Control | 1 Tube of ASF Inhibition Control |
| Product Insert | 1 Product Insert |
Workflow Summary: PCR Workflow (See Product Insert for Complete Details)

To Order
-
- Purchase restrictions may be applicable. Contact us and reference catalog number TC-9096-096.
Additional information
| All | All |
|---|
1 review for VetAlertTM African Swine Fever Virus (ASFV) DNA Test Kit
You must be logged in to post a review.


Zach Polhemus –
great